I. Introduction
September marks the 5th Anniversary of both the America Invents Act (AIA) and the birth of the Inter Partes Review (IPR). Upon its advent, the life sciences patent bar eagerly pondered if and how IPRs would be used. After all, Hatch-Waxman had been the centerpiece of life sciences litigation. After about five years of IPR experience under our belts, we can now see if and how IPRs have replaced, interplayed with, or conflicted with traditional Hatch-Waxman litigation.
For several decades, Hatch-Waxman district court litigation was the forum du jour for innovators and generics to fight their battles. After much debate from players in the industry, Congress crafted the Hatch-Waxman mechanism. Lawmakers sought to address the competing interests of an innovator’s patent rights and a generic drug company’s ability to potentially offer a cheaper version of a drug. To strike a balance, Congress provided successful generic challengers with a 180-day market exclusivity and innovators with a 30-month stay of FDA approval of any generic product. Although complex, the Hatch-Waxman framework has generally served its purpose well since its enactment in 1984 with subsequent amendments.
Yet, in 2012 the AIA established an alternate mechanism to challenge patent validity via the IPR. This article contemplates trends involving generics, IPRs and Hatch-Waxman district court litigation.
II. IPRs: Where Are We Now?
IPRs provide an alternate forum for generics to invalidate pharmaceutical patents because they may perceive IPRs to be a more favorable proceeding. For example, as opposed to district court litigation, IPRs allow only limited discovery and apply a lower burden of proof, a preponderance of the evidence standard. Also, grounds for challenging validity in an IPR are limited to novelty and obviousness based upon printed publications. Therefore, IPRs provide a comparably affordable venue for patent challengers. Patent challengers in all technology sectors have taken advantage of IPRs, including in the life sciences area. However, petitions filed in the life sciences area are dwarfed by those filed in the other technology areas.
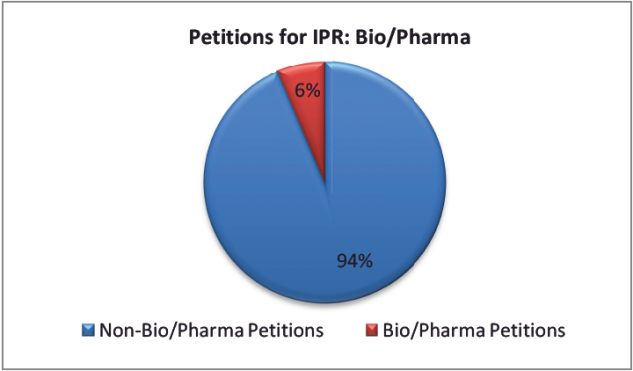
Thus far, over 6,700 petitions for IPRs have been filed. Although filings of petitions for IPR in all technology sectors are on the rise, petitions targeting pharmaceutical patents remain a small percentage of total petitions.
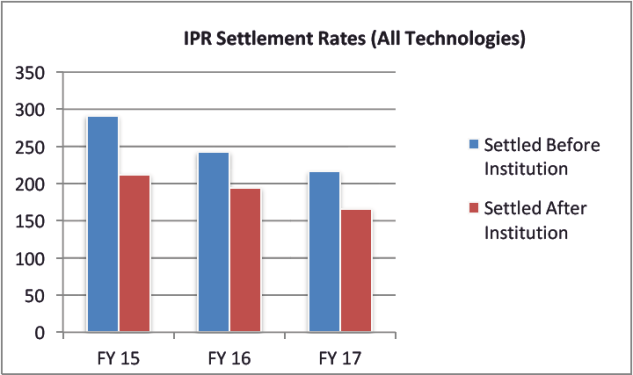
Notably, the initial fear of the Patent and Trademark Appeal Board (PTAB) as the firing squad is not necessarily coming true in the pharmaceutical context. Of the more than 6,700 petitions filed, only about 500 involve bio/pharma patents. The PTAB has instituted trials for about 63% of bio/pharma petitions and has found about 50% the claims at issue unpatentable. There tends to be a higher settlement rate for IPRs of pharmaceutical patents.
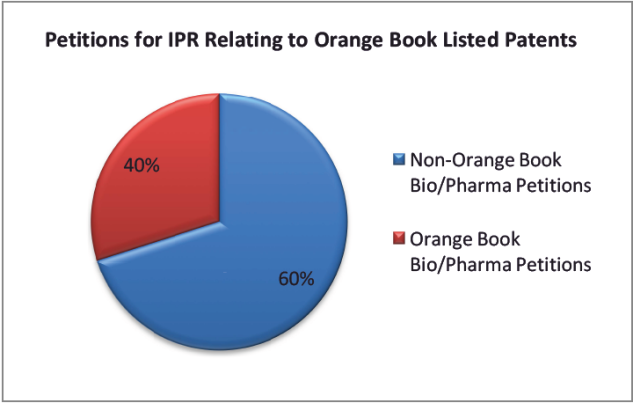
Interestingly, since the creation of the AIA about 200 of the 500 bio/pharma petitions that have been filed involve patents listed in the Food and Drug Administration’s Orange Book, covering about 120 drugs. Of those petitions, about 130 have been instituted.
A majority of the instituted IPRs that involve an Orange Book patent are still pending or have been terminated. For IPRs where the PTAB has issued a final written decision, it has cancelled claims in about half those instances.
Overall, IPRs involving Orange Book patents are less likely to be instituted than IPRs involving other technology. It is also less likely that all of the claims involved will be invalidated.
III. IPRs and Hatch-Waxman Disputes
Generic companies include IPRs in their strategy. Perhaps, because IPRs may be an easier path to obtain market entry. The tactic of using IPRs in for Orange Book listed patents has been adopted by numerous generic pharmaceutical companies. These companies include Amneal Pharmaceuticals, Mylan Pharmaceuticals, Agila Specialties, Akorn, Inc., Alembic Pharmaceuticals, Amerigen Pharma, Torrent Pharmaceuticals, Apotex Inc., Teva Pharmaceuticals, Accord Healthcare, Sandoz Inc., Neptune Generics, Argentum Pharmaceuticals, Aurobindo Pharma, Breckenridge Pharma, Roxane Laboratories, Par Pharmaceutical, Dr. Reddy’s Laboratories, Endo Pharmaceuticals, Fresenius Kabi, Lannett Holdings, Lupin Limited, InnoPharma Inc., Panacea Biotec, Noven Pharmaceuticals, Actavis Pharmaceuticals, and Sawai USA Inc.
A generic company can use IPRs in different ways to position itself to enter the market as early as possible. There is no specific timeframe as to when a company can challenge a patent through an IPR. Thus, a generic can file an IPR before the expiration of the four-year waiting period that exists under the Hatch-Waxman structure if a product has New Chemical Entity (NCE) exclusivity. Although this option may expedite resolution of patent issues that present barriers to a generic pursuing a product, it may not be the first choice of many. If a generic removes the patent barriers to entry before the expiration of the four-year waiting period, it may result in the removal of its ability to qualify for the 180-day exclusivity. But this avenue cannot be discounted and an innovator company, therefore, should consider this possibility. Add into the calculus the fast pace at which IPRs progress, and it may become more of an issue that an innovator may not enjoy the full period of exclusivity.
The interplay between the 180-day exclusivity and IPRs does not end at whether it would deter a generic company from challenging a patent before filing of an Abbreviated New Drug Application (ANDA). A second generic seeking to invalidate a patent that forms the basis of a first filer’s 180-day exclusivity may file an IPR. If successful, the second generic could seek to use the IPR decision either with a district court or through affirmance at the Federal Circuit as a triggering event. Often times, multiple patents are listed in the Orange Book for a product. This allows a generic to pick and choose the venue where it challenges each Orange Book listed patent. If a generic company can develop a strong non-infringement position for one of the listed patents, it can challenge that patent in a district court litigation to obtain the 180-day exclusivity. A generic company can then challenge the validity of any patent for which it does not have a strong infringement defense at the PTAB. This will offer the benefit of a different claim construction standard that lends itself to broader claim constructions than may be obtainable in a district court. See, e.g., PPC Broadband, Inc. v. Corning Optical Communications RF, LLC,
First filers may not be the only generic companies that will use IPR proceedings. A likely scenario could involve generic companies that do not qualify for the 180-day exclusivity. They can seek to use an IPR as a strategy to trigger a first filer’s exclusivity. Although an invalidation at the PTAB will not qualify as a triggering event, it may provide a second filer an opportunity to convert that decision into a trigger event through a successful appeal or use of that decision to convince a district court to issue a decision in favor of the second filer.
IV. How IPRs Have Played Out in the Hatch-Waxman Context
It still seems that the primary locus of activity of Hatch-Waxman disputes has, and will remain, the district courts. At present, generic companies are challenging over 1,000 Orange Book listed patents in district courts. However, several of these patents are the subject of parallel IPR proceedings at the PTAB.
A. Generics Prevail at the PTAB
The option for a generic challenger to file an IPR exposes innovators to the fact that they may have to defend their patents on two battle-fronts. This fact, in certain instances, has allowed generic challengers to effectively override a patent owner’s choice of forum in ANDA disputes, and instead litigate the disputes in forums more favorably disposed to validity challenges. This appears to be the case with challenges to Eli Lilly and Daiichi Sankyo’s Effient® franchise. The Orange Book identifies three patents—U.S. Patent No. 5,288,726, U.S. Patent No. 8,404,703, and U.S. Patent No. 8,569,325—as protecting this franchise. With pediatric exclusivity, these patents expire on October 14, 2017, July 2, 2023, and July 2, 2023, respectively. On March 12, 2014, Eli Lilly filed a complaint asserting these patents against multiple generic challengers in the Southern District of Indiana. The next day, Eli Lilly filed a suit in New Jersey, presumably as a protective suit, in case jurisdiction was challenged in Indiana. In March 2015, several of the generic challengers submitted IPR petitions challenging the patentability of U.S. Patent Nos. 8,404,703, and 8,569,325. The PTAB instituted trials against these two patents. Following that institution, some of the generic challengers requested a stay of the district court proceeding and the district court granted that stay. In its order, the district court rejected the patent owner’s request that the stay be conditioned on an extension or tolling of the 30-month stay associated with the district court litigation. The PTAB issued a final written decision in both IPRs in September 2016 in which it cancelled the claims of both patents. Upon notifying the district court of the IPR results, the district court maintained the existing stay pending the appeal of the matter. By using the IPR process, the generic challengers in this matter effectively overrode the innovator’s choice of forum and had the parties litigate this matter in a forum more favorable to the generic challengers.
There are examples where, unlike the example above, a district court has decided to proceed despite the pendency of IPRs against the patents forming the basis of the district court litigation. In Hatch-Waxman litigations related to the drug Copaxone®, Judge Gregory M. Sleet of the District of Delaware continued the district court litigation even after the PTAB found several of the claims that formed the basis of the lawsuit unpatentable. The district court trial involved U.S. Patent Nos. 8,232,250, 8,399,413, 8,969,302, and 9,155,776. In February and March of 2015, about five months after the litigation began, Mylan submitted IPR petitions against U.S. Patent Nos. 8,232,250, 8,399,413, and 8,969,302. U.S. Patent No. 9,155,776 issued in October 2015 and was added to the existing litigation. Mylan then submitted a post grant review petition against U.S. Patent No. 9,155,776. In its final written decisions of August and September 2016, the PTAB held the claims of U.S. Patent Nos. 8,232,250, 8,399,413, and 8,969,302 unpatenable. Despite the PTAB’s decision, Judge Sleet conducted a seven-day bench trial at the end of September 2016. In January 2017, Judge Sleet issued his decision and held the asserted claims of U.S. Patent Nos. 8,232,250, 8,399,413, 8,969,302 and 9,155,776 invalid. Judge Sleet issued a lengthy decision in which he explained his bases for finding the claims invalid for obviousness. In that decision, Judge Sleet’s reference to the PTAB result was sparse. In a footnote at the end of his opinion, Judge Sleet recognized that the PTAB had come to similar conclusions about the lack of patentability of the asserted claims. Judge Sleet noted that if the patent owner appeals both the IPR decisions and his ruling, it will present an interesting procedural posture for the Federal Circuit. In essence, the cases are identical but subject to different standards in analyzing the patentability of the claims. The Copaxone® cases are of interest because they show that district courts may be unwilling to simply rubber-stamp a PTAB conclusion that a claim is not patentable, considering it is charged with analyzing the claims under a different standard.
But in general, one party’s success in an IPR will likely benefit other generic companies because the other companies may use that success to prevail in Hatch-Waxman litigation. Alembic Pharmaceuticals filed a complaint in March 2017 seeking a declaratory judgment of non-infringement and invalidity of U.S. Patent No. 8,324,283 in the Eastern District of Virginia. Alembic Pharmaceuticals v. Novartis, Case No. 1-17-cv-00292 (E.D. Va. June 13, 2017). The patent at issue is owned by Novartis and Mitsubishi Tanabe Pharma, and covers Novartis’ product, Gilenya®. Novartis and Mitsubishi did not sue Alembic over its paragraph IV notice. In its complaint, Alembic cited a successful IPR decision filed by another generic that related to the ʼ283 patent. In that final written decision, the PTAB found all the asserted claims of the ʼ283 patent unpatentable. The decision was appealed and was being considered by the Federal Circuit when Alembic filed the declaratory judgment action. Shortly after bringing suit, the Federal Circuit affirmed the PTAB’s decision. Thereafter, the district court granted Alembic the declaratory judgment that the ʼ283 patent was invalid and, thus, the proposed generic product would not infringe. The strategy to file suit while the PTAB’s decision was on appeal, and, therefore, not final, guaranteed the challenger jurisdiction. This case also poses interesting questions of how IPR decisions can be enforced to affect Hatch-Waxman litigation.
B. Generics Lose at the PTAB
Unsuccessful efforts by parties in IPR proceedings also teach valuable lessons. In Ino Therapeutics v. Praxair Distribution, Ino Therapeutics brought a Hatch-Waxman suit in the District of Delaware alleging that any generic version of its INOmax® product would infringe its valid patents. Case No. 1-15-cv-00170 (D. Del. September 5, 2017). Concurrently, Praxair used IPR proceedings to challenge Ino’s patents at issue. The PTAB instituted some petitions, but denied others. IPR2015-00522; IPR2015-00524, IPR2015-00525, IPR2015-00526; IPR2015-00529; IPR2015-00891; IPR2015-00893; IPR2015-00884; IPR2015-00888; IPR2015-00889. In its IPR petitions, Praxair argued that the patents were invalid as obvious under
Failure to achieve victory through an IPR proceeding is not necessarily a death knell to a generic company’s district court chances, but that effectively may be the result. A review of ANDA submissions related to the Lexiva® franchise provides an example of such a result. The Lexiva® franchise was protected by two patents—one that claimed the active ingredient and a second that claimed a polymorphic form of the active ingredient. Perhaps realizing that any generic product necessarily would infringe the latter patent, two generic companies filed an IPR on that patent. The first generic challenger was able to convince the PTAB to institute an IPR on all the claims of the compound patent and that IPR was resolved without issuance of a final written decision. A second generic challenger sought to leverage the institution decision that the first generic challenger received by submitting a copycat petition. However, the PTAB denied institution of several claims of the compound patent in light of evidence the patent owner had developed during the first IPR proceeding. Seeking to rectify this issue, the second generic challenger submitted a revised petition against the claims for which the PTAB had denied institution. Through the submission of this petition, the second generic challenger was able to achieve institution on the earlier excluded claims. In the end, however, the PTAB issued a final written decision concluding that the second generic challenger failed to prove the claims unpatentable. See Lupin Limited v. Vertex Pharmaceuticals Inc., IPR2016-00558 (Paper 39). Because of the timing of this decision, the generic challenger effectively ran out of time to meaningfully challenge this patent at the district court. Even though the IPR process offers a powerful weapon to generic challengers, it must be used carefully. If a generic challenger can only muster a prior art attack on a patent, then a loss before the PTAB may effectively end the challenger’s ability to achieve a win at the district court that may be important for approval or exclusivity reasons.
V. Conclusion
At its five year mark, the AIA and its star player, the IPR, have raised more questions relating to their roles in the life sciences field and in Hatch-Waxman litigation. Although we have only started to see how and to what extent IPRs shape Hatch-Waxman litigation, there are key takeaways from observing the new proceedings thus far.
Firstly, Orange Book patents make up a fraction of the asserted patents in the total number of IPR petitions. Bio/pharma petitions are being instituted at a rate of only approximately 64% and only about half of the claims asserted are being cancelled in IPRs. Additionally, there is a higher settlement rate for Orange Book patents. Orange Book patents are also less likely to be instituted or invalidated than other technology sectors.
Secondly, five years of the AIA has demonstrated the various strategies employed by using IPRs to affect Hatch-Waxman litigation. Generic pharmaceutical companies may file IPRs prior to expiration of NCE exclusivity period as an offensive weapon. However, these generics would most likely be deterred by the possible loss of 180-day exclusivity eligibility. Though it is a risk, innovators should be aware that it is a powerful play if successful. Second filers could also file IPRs in an attempt to capture the market.
Generic companies have also been able to use IPRs to pick and choose a preferred forum. By taking a fight to the PTAB, the generic may benefit from a lower evidentiary standard and a different claim construction standard.
Thirdly, both innovators and generics could learn from the mixed reactions of the courts to concurrent IPRs or IPRs that consider the patents that form the bases of the district court litigation. Some courts have effectively denied the patent owner its preferred forum by staying cases pending the IPR. Other courts have proceeded with a case to make their own determinations, disregarding the cancellation of claims at the PTAB. In one instance, a generic won a Hatch-Waxman litigation by raising a PTAB decision that was subsequently affirmed by the Federal Circuit to serve as the basis for a declaratory judgment of invalidity.
Finally, it is notable that failure at the PTAB also has its implications in concurrent or subsequent Hatch-Waxman litigation. An unsuccessful IPR may leave generic companies vulnerable in parallel or subsequent district court litigation by revealing their offensive strategies or exhausting their options.
In conclusion, the majority of life sciences litigation will remain in the district courts firmly within the Hatch-Waxman framework. However, the implications of IPR proceedings must not be underestimated.
Learn more about Bloomberg Law or Log In to keep reading:
See Breaking News in Context
Bloomberg Law provides trusted coverage of current events enhanced with legal analysis.
Already a subscriber?
Log in to keep reading or access research tools and resources.