The Environmental Protection Agency is authorized through statutes such as the Comprehensive Environmental Response, Compensation, and Liability Act and others to develop action thresholds for both residential and commercial worker exposures. Similarly, the Occupational Safety and Health Administration is authorized to protect workers from significant risks in the workplace for which the agency develops its own regulatory action thresholds. There is often considerable confusion and overlap regarding whether EPA or OSHA has authority at a given workplace.
Many other agencies are also authorized to develop these action thresholds. The Agency for Toxic Substances and Disease Registry (ATSDR) develops non-cancer short- and long-term exposure thresholds that overlap with EPA long-term non-cancer thresholds. The National Toxicology Program (NTP) is a “virtual agency” comprising the National Institute for Environmental Health Sciences (NIEHS) and the National Institute for Occupational Safety and Health (NIOSH) within the Centers for Disease Control and Prevention (CDC) and the National Center for Toxicological Research within the Food and Drug Administration, conducting studies to determine the toxicity of specific chemicals and to develop and validate study methods.
The assessment of risk and decisions regarding acceptable risk fall under a process generally termed “risk assessment.” In practical application, risk assessment consists of three distinct functions, risk assessment, risk management and risk communication. Risk assessment and risk management rely significantly on assumptions and judgments. In brief, risk is a function of both exposure and toxicity. Either longer exposure times, increased exposure concentrations or designating a chemical as having greater toxicity will result in increased risk. Thus, development of health protective exposure levels or standards requires assessments of both exposure and toxicity. Different agencies make different assumptions about exposure and toxicity, with little to no coordination of results or overlap.
For instance, at a hazardous waste site cleanup where there is indoor air, soil or groundwater exposure, the risk assessor developing acceptable exposure levels often does not really track a person’s daily activities and, therefore, they do not know the exact exposure conditions for each person in the impacted area. Instead, they make conservative assumptions regarding exposure time, frequency and duration and apply these assumptions to the population under consideration. This same subjective judgment also governs the assessment of the toxicity of a given contaminant. Generally, animal studies are used to determine toxicity, and results are extrapolated to humans. The extrapolation requires subjective judgment, which is not similar among agencies. For instance, ATSDR may have a different toxicity assessment than EPA’s cleanup programs (e.g., CERCLA, Resource Conservation and Recovery Act) or its Toxic Substances Control Act program (see Table 1). Because these uncertainties exist for every step of the risk assessment and management process, scientists and risk managers must make assumptions and judgments that significantly impact the results for action levels or standards.
A respective agency’s guidelines for the development of protective thresholds are structured such that considerable professional judgment is required. Thus, each agency or regulatory body that develops such thresholds exercises considerable professional judgment, and each differing threshold is treated by the respective regulatory body as being the “correct one.” While consummate professionals in the field understand these discrepancies and overlaps, the common user does not, and trying to determine who has regulatory authority is often maddening. Given the government’s lack of resources,
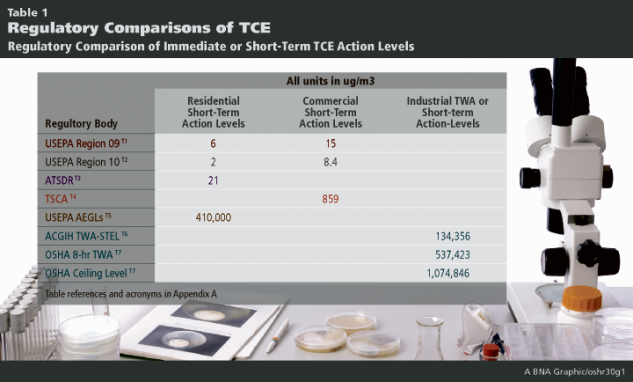
An example of the failure of multi-agency standard-setting is found in the development of indoor air action levels for trichloroethylene (TCE). OSHA, EPA, ATSDR, EPA’s TSCA program, EPA’s regional offices and many states have developed indoor air action thresholds that are remarkably different. These action thresholds are applied to short-term exposures. In general, thresholds are developed for both long-term or chronic exposures and shorter-term or immediate exposures. The table below illustrates these differences for short-term exposures and is thus labeled short-term action levels.
It is apparent that there is considerable and significant disagreement among differing agencies and their respective residential, commercial or industrial exposure action levels. It should be noted that, unlike other action thresholds, the OSHA Time Weighted Averages are Permissible Exposure Limits (PELs) and as such are legally enforceable standards. The other action thresholds, which trigger investigation or remedy, are often called standards, but are thresholds that would trigger necessary action to avoid exposures at “unsafe” levels, and in practice these are not legally enforceable per se. Likewise, state efforts are inconsistent and add an additional layer of confusion to this important public health issue (Inside EPA, 2012; USEPA Region 10, 2012; New Hampshire, 2013). Perhaps the most glaring concern is that the government is sending multiple, confusing messages about TCE inhalation exposure risks, especially short-term action levels.
EPA and others have applied the most recent short-term action level as given in the table in an attempt to account for the potential occurrence of developmental health effects (fetal cardiac malformations) for women exposed to TCE during pregnancy. In its controversial decision, EPA (2011) relies upon three studies. Two of these studies (Kiel et al. 2009; NTP, 1988) show no short-term or developmental effects. However, the third study used by EPA, (Johnson et al., 2003), does measure short-term developmental effects of oral doses of TCE to mice.
EPA’s Risk Assessment Guidance for Superfund (RAGS, 1989) and its Guidelines for Developmental Toxicity Risk Assessment (EPA, 1991) provide guidance for EPA’s derivation of a developmental inhalation reference concentration (RfC) and the application of the Johnson study to develop a chronic RfC that is also applied as a stand-alone inhalation developmental RfC is difficult to support given principles discussed in these guidance documents. This third study is used to evidence the risk of fetal heart malformations. However, this study was not an inhalation study. The researchers gave oral doses (i.e., not inhalation doses) of the chemical to the animals, and the oral doses were extrapolated to inhalation exposures through the use of probability-based models. Importantly, its results have not been reproducible by other studies and inhalation studies have not detected such a risk.
By comparison, ATSDR recently performed two health consultations (2013, 2013a) using the health effects level, 21 µg/m3, from the EPA TCE Toxicological Review (2011) as an intermediate exposure duration (14-365 days, less than one year) action level. ATSDR compared this value to estimated or measured daily 24-hour exposure levels in order to make recommendations on the risk of a toxic effect.
Even more controversial and demonstrative of the free range of regulatory professional judgment is the difference between the TSCA program and the U.S. EPA regions’ standards. Here the EPA regions have used a single oral study that was “route” extrapolated from oral to inhalation through the use of sophisticated biological modeling. In contrast, EPA’s TSCA program, in its draft Workplan Chemical Risk Assessment for Trichloroethylene (2012), used only inhalation studies, citing the fact that the toxic effect seen in the oral study is not evident in inhalation studies as reasonable justification. This difference is remarkable considering that the TSCA program is a part of EPA. As suggested by the table above, not only does the TSCA program disagree, but ATSDR and others also do not agree with the EPA regions’ short-term action level. It is recognized in the scientific community that toxicity by a given exposure route (e.g., inhalation, ingestion, etc.) may not operate the same, and, therefore, chemical risks and toxic effects associated with ingestion of a chemical do not necessarily mean a similar toxic effect or risk will result from inhalation exposure.
This real-life scenario is problematic for a number of reasons. First, the EPA regions’ new short-term action level may have significant impacts in the community as detection of TCE at or above the action level will trigger high-priority, time-critical, rapid actions, such as the potential evacuation of a building. This issue has not escaped the eye of stakeholders who provided comments to EPA regarding the draft Office of Solid Waste and Emergency Response vapor intrusion guidance (“VI Guidance”) released by EPA for public comment April 15, 2013:
“We urge EPA to fully evaluate the potential economic disruption and fear that its approach to short term action levels may generate, to consider the substantial uncertainty in the science that underlies the development of short-term action levels and the most recent IRIS reference concentration for TCE, and to develop short-term levels that [are] consistent with the science and reasonable.”
The EPA regions’ new short-term action level sets up a complicated dichotomy of government regulation. The critical effect, according to EPA Regions 9 and 10, is fetal heart malformations in the developing fetus. By contrast, the TSCA program cites the critical effect as neurotoxicity and does not address or otherwise describe any association with fetal heart malformations. OSHA takes a position similar to the TSCA program, in that they do not cite any developmental fetal heart malformations as a critical effect. Thus, EPA Regions 9 and 10 will regulate and may remove pregnant women in the workplace from exposure at 8-15 µg/m3, while OSHA will not remove a worker (pregnant or otherwise) from exposure less than 540,000 µg/m3, about a 50,000-fold difference. This is simply unacceptable. How can our science body be so far apart on this issue? Here is a hypothetical provided by a stakeholder submitting comments to EPA for the VI Guidance:
“The average TCE concentration in a workplace’s breathing air is 500,000 µg/m3. No response may be required by EPA. Why? Though EPA now calculates that workers are exposed to unacceptable cancer risks at levels above 3 µg/m3 over years of exposure—and some EPA regions have concluded that developing fetuses are at risk of heart malformations if pregnant women are exposed above 5 µg/m3 for even 24 hours and would evacuate workers from workplaces at 15 µg/m3—TCE is used in this workplace, and workers have access to a Material Safety Data Sheet explaining its risks under OHSA rules.”
So, pregnant women in the occupational setting regulated by OSHA and exposed to the higher concentrations are fine, but in settings regulated by EPA, the agency may evacuate the building (or an area within the building) at levels approximately 50,000 times lower. Really? Does EPA’s reliance upon a controversial study, where the scientists extrapolate from oral to inhalation exposures and risks, simply create scientific uncertainty, public confusion and fear? Some experts are further frustrated, alleging that EPA’s risk assessment process lacked transparency. Perhaps CH2M Hill’s comments are most relevant to this article:
“A credible and transparent process is needed to assure that values for assessing short-term exposures are consistent across [US]EPA programs such as the cleanup programs (Comprehensive Environmental Response, Compensation, and Liability Act [CERCLA] and Resource Conservation and Recovery Act [RCRA] Corrective Action) and toxics programs (under Toxic Substances Control Act [TSCA]), and consistent across [US]EPA regions and other federal agencies including the Agency for Toxic Substances and Disease Registry (ATSDR) and U.S. Occupational Safety and Health Administration (OSHA). If [US]EPA chooses to retain the section of the [Final Vapor Intrusion Guidance] on short-term exposure and rapid action levels, the methodologies to ‘identify health-protective concentration levels for vapor-forming chemicals based upon potential noncancer health effects that can be posed by air exposures over short-term or acute exposure durations’ need to be included and based on peer-reviewed, defensible approaches.”
We are discussing human health risk for the inhalation of TCE vapors and the impact on pregnant women. If there is such a problem, then the government should SPEAK WITH ONE VOICE. Wouldn’t it be better for one agency to collaborate with the other agencies and determine what the action level should be?
Why is this necessary? The scientific community has stated that consistency, reproducibility and fairness are important goals in the risk assessment process.
“[R]isk assessment is at a crossroads. Despite advances in the field, it faces a number of substantial challenges, including long delays in completing complex risk assessments, some of which take decades to complete; lack of data, which leads to important uncertainty in risk assessments; and the need for risk assessment of many unevaluated chemicals in the marketplace and emerging agents.”
Whether the agency is analyzing the short-term or chronic (e.g., long-term) effects of chemical exposure, it is a difficult task. Variability,
The well-documented complexities inherent within risk assessment must be faced and resolved. However, it is inevitable that the creation of legal frameworks where multiple agencies set regulatory action thresholds using a process overly reliant upon multiple agencies’ differing assumptions and judgment would naturally be subject to disagreements in the application of the science. But, when it comes to a government determination about how much chemical may be safely inhaled, ingested, etc., any standard should be, in the words of the Committee on Improving Risk Analysis Approaches Used by the U.S. EPA, consistent, reproducible and fair.
The agency could set action levels and/or standards for all exposures by media. If an action level or standard requires adjustment to account for differences in an occupational environment (e.g., the amount of time at work), then that agency should be able to make the adjustment based on the same risk assessment framework and explain how and why it adjusted its level for the workplace. Thus, deviations from prior risk assessments would be fully explained and justified to affect sufficient peer review. As stated by the National Research Council years ago, transparency in standard-setting is critical.
The best way to enable consistency in a process so fraught with differing judgments and assumptions is to place the public health (e.g., risk assessments) and environmental chemical exposure (e.g., ecological assessments) action level or standard setting in the hands of one agency, perhaps a Standards Bureau, and to ensure that agency establishes a transparent, inclusive process consistent with known science policy. This would also minimize, though not eliminate, the potential for these assumptions and judgments to be tainted with scientific and agency biases and politics.
A classic example of alleged bias can be found in the infamous EPA Secondhand Smoke Study from 1993.
Unrelated to the passive smoking case, the National Research Council wrote in a report years ago: “In summary, informed and, in some cases, ground-breaking governance are intended to improve USEPA risk-assessment processes, focus the assessment on the relevant questions, discourage political interference or pre-determined policy biases, and promote senior-level oversight of the timeliness, relevance, and impact of decision-making.”
In 2013, members of three professional societies
John S. Applegate of the Indiana University Maurer School of Law wrote in A Beginning and Not an End in Itself, “In an ideal world, scientists and the public would have exact, complete and uncontested information on the factors responsible for pollution-related environmental and human health problems. … [I]n that ideal world time and resources would not be limiting, and the requisite information and analyses would be available at the time needed, and in the quality and quantity needed, so that decision makers would be able to make decisions consistently using relevant data.”
Appendix A: Table References and Acronyms
1. USEPA Region 9: Information taken from Inside USEPA Superfund Report Volume XXVI, No. 12—June 11, 2012 and TCE Interim Short-Term Removal Action Level White Paper prepared by Exponent and Geosyntec, April 17, 2012. USEPA Region 9 used the RfC at 2.0 µg/m3 and multiplied it by a 24/10 factor to simulate 10-hour commercial work day, rounded 4.8 to 5 µg/m3 and then multiplied 5 µg/m3 by the recommended factor of 3 from the USEPA 2008 Remedial Action Level (RAL) Memo. In the USEPA RAL Memo an HI of 3 for removal actions is used. The residential immediate action level of 6 µg/m3 was derived similarly for comparison by using the RAL memo recommendation for an HI of 3 x 2 µg/m3.
2. USEPA Region 10 values taken from Dec 12, 2012 Memorandum: “OEA Recommendations Regarding Trichloroethylene Toxicity in Human Health Risk Assessments.”
3. ATSDR used the HEC99 from USEPA 2011 Toxicological Review of Trichloroethylene (USEPA, 2011) in both the Millsboro Consultation (ATSDR, 2013) and the Pohatcong Valley Consultation (ATSDR, 2013a).
4. TSCA: Data taken from TSCA Workplan Chemical Risk Assessment for Trichloroethylene: Degreaser and Arts/Crafts Uses CASRN: 79-01-6 Ethene, 1,1,2-trichloro (Draft). This is considered a “light commercial exposure.” Short-Term screening value derived using equation page 60 and acute HEC99 from Table 3-21.
5. Acute Exposure Guideline Level (AEGL) information from http://www.epa.gov/oppt/aegl/pubs/define.htm is the airborne concentration, expressed as parts per million or milligrams per cubic meter (ppm or mg/m3) of a substance above which it is predicted that the general population, including susceptible individuals, could experience notable discomfort, irritation, or certain asymptomatic non-sensory effects. However, the effects are not disabling and are transient and reversible upon cessation of exposure. TCE has Interim AEGLs 77 ppm is most sensitive of all the AEGL categories. Note that “AEGLs are intended to describe the risk to humans resulting from once-in-a-lifetime, or rare, exposure to airborne chemicals.”
6. American Council of Governmental Industrial Hygienists Short Term Exposure Levels. ACGIH STEL is short term exposure limit from 2010 ACGIH published values. This value agrees with or less than NIOSH 10 hr. TWA see: http://www.cdc.gov/niosh/npg/nengapdxc.html.
7. OSHA 8-hour Time Weighted Average (TWA) and Ceiling Value available at: https://www.osha.gov/dts/chemicalsampling/data/CH_273000.html last accessed 4/15/14.
Appendix B: Additional References
ATS
ATS
Alliance for Risk Assessment, 2013 Guidance for Contaminated Sites: Trichloroethylene (TCE) Risk Assessment Case Study available at:http://www.allianceforrisk.org/
DOD, 2011 Department of Defense Comments on TCE IASD final draft Tox Review Chemical Material Risk Management Directorate Department of Defense 7/15/11
Hardin et al, 2004 Bryan D. Hardin, Kelman, Bruce J., Brent, Robert L. Trichloroethylene and Cardiac Malformations. Environ Health Perspect. Aug 2004; 112(11): A607–A608.
Inside EPA, 2012 Superfund Report Vol XXVI No. 12. June 11, 2012. www.inside.epa.com “EPA Weighs Short-Term Clean-up Limit for TCE Worrying Industry”
Johnson et al, 2003 Johnson, P; Goldberg, S; Mays, M; Dawson, B (2003). Threshold of trichloroethylene contamination in maternal drinking waters affecting fetal heart development in the rat. Environ Health Perspect 111: 289-292.
Kiel et al 2009 Keil, D; Peden-Adams, M; Wallace, S; Ruiz, P; Gilkeson, G (2009). Assessment of trichloroethylene (TCE) exposure in murine strains genetically-prone and non-prone to develop autoimmune disease. J Environ Sci Health A Tox Hazard Subst Environ Eng 44: 443-453. http://dx.doi.org/10.1080/10934520902719738
New Hampshire, 2013. The State of New Hampshire, Department of Environmental Services, Thomas S. Burack, Commissioner, February 7, 2013 Waste Management Division Update: Revised Vapor Intrusion Screening Levels
NTP, 1988 NTP. (National Toxicology Program). (1988). Toxicology and carcinogenesis studies of trichloroethylene (CAS No. 79-01-6) in four strains of rats (ACI, August, Marshall, Osborne- Mendel) (gavage studies). Research Triangle Park, N.C.: U.S. Department of Health and Human Services, Public Health Service, National Institutes of Health. http://ntp.niehs.nih.gov/ntp/htdocs/LT_rpts/tr273.pdf
RAGS, 1989 Risk Assessment Guidance for Superfund, Volume 1, Human Health Evaluation Manual (Part A) EPA/540/1-89/002, December 1989.
USEPA, 1991 Guidelines for Developmental Toxicity Risk Assessment EPA/600/FR-91/001 December 1991
Published on December 5, 1991, Federal Register 56(234):63798-63826 Risk Assessment Forum
U.S. Environmental Protection Agency Washington, D.C.
USEPA 2011 Toxicological Review of Trichloroethylene In Support of Information on the Integrated Risk Information System, September, 2011. EPA/635/R-09/011F www.epa.gov/iris
USEPA Region 10, 2012 Memoradum: OEA Recommendations Regarding Trichloroethylene Toxicity in Human Health Risk Assement From: Joyce C. Kelly Director, To: Rick Albright and Kate Kelly, Dec. 13, 2012
Learn more about Bloomberg Law or Log In to keep reading:
See Breaking News in Context
Bloomberg Law provides trusted coverage of current events enhanced with legal analysis.
Already a subscriber?
Log in to keep reading or access research tools and resources.